The First Generation of Programmable Genome Editors
Long before CRISPR and even before TALENs, zinc finger nucleases (ZFNs) marked the beginning of truly programmable genome editing. Their development demonstrated for the first time that scientists could design proteins capable of recognizing specific DNA sequences and inducing targeted genetic changes. This breakthrough laid the foundation for the genome engineering revolution that followed, and although newer technologies have surpassed ZFNs in simplicity and scalability, their historical and scientific importance remains undeniable.
How Zinc Finger Proteins Recognize DNA
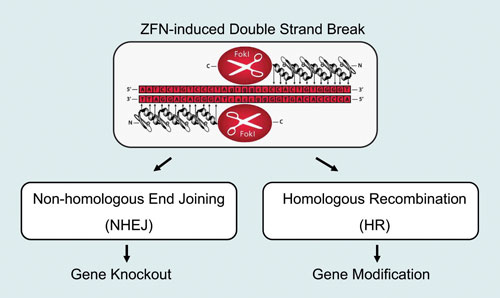
ZFNs are built from zinc finger proteins—small, modular DNA-binding motifs that each recognize a triplet of nucleotides. By assembling multiple zinc fingers in tandem, researchers can create a protein capable of binding long, unique DNA sequences with high specificity. This modular design was a major conceptual leap, proving that DNA-binding proteins could be engineered rather than discovered. As described in the presentation, this ability to customize DNA recognition was one of the earliest demonstrations of targeted genome manipulation.
FokI Nuclease: The Engine Behind the Edit
While the zinc finger domains provide the targeting capability, the actual DNA cleavage is performed by the FokI nuclease domain. Like TALENs, ZFNs operate as dimers: two ZFN constructs bind opposite strands of DNA, bringing their FokI domains together to create a double-strand break. This break activates the cell’s repair pathways, enabling gene knockout, insertion, or correction. Although designing ZFNs requires significant expertise and labor, their precision and reliability made them indispensable tools in early genome editing research.
ZFNs in Modern Biotechnology
Even with the rise of CRISPR, ZFNs continue to play a role in therapeutic development and industrial biotechnology. They have been used in clinical trials, including some of the earliest gene-editing therapies for HIV, and remain valuable in contexts where regulatory familiarity or specific design constraints favor their use. The presentation underscores their pioneering status, reminding us that today’s genome editing landscape was built on the innovations introduced by ZFNs.
Image source: https://www.genengnews.com/wp-content/uploads/2018/10/UGENWebsitepictures2010GEN13_July10AssayTutorialSigmaSigma_Fig11863122711.jpg