The Test That Makes Precision Medicine Possible
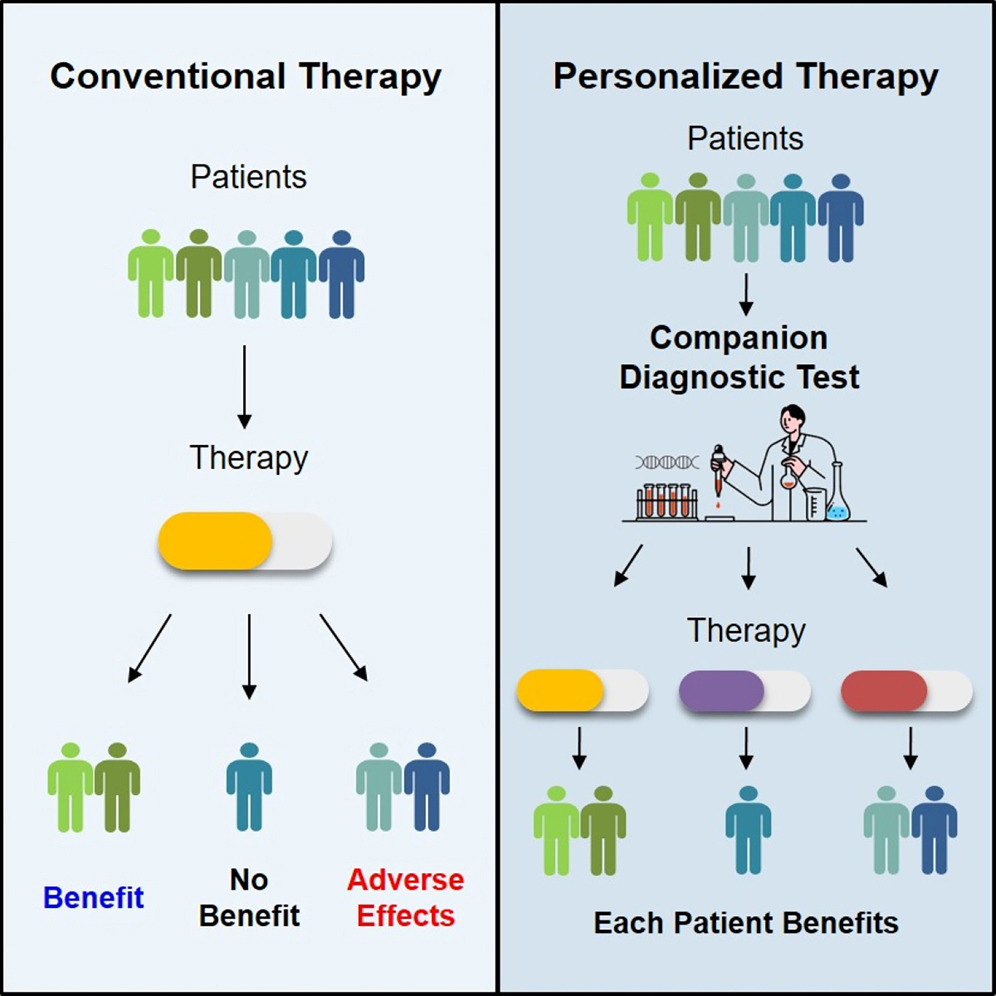
Precision medicine begins long before a therapy is prescribed. It starts with the diagnostic test that determines whether a patient is likely to benefit from a specific treatment. Companion diagnostics (CDx) have become the backbone of targeted therapy, ensuring that the right patients receive the right drugs at the right time. As targeted treatments expand across oncology, immunology, and rare diseases, CDx is emerging as the essential gatekeeper of therapeutic success.
Understanding the Power of Biomarker‑Driven Care
Companion diagnostics work by identifying biomarkers—genetic, protein‑based, or molecular signatures—that predict how a patient will respond to a therapy. These tests guide clinicians in selecting treatments that are more likely to be effective while avoiding those that may cause harm or offer no benefit. This approach transforms treatment from a generalized protocol into a personalized strategy, improving outcomes and reducing unnecessary toxicity.
A New Era of Targeted Therapies
The rapid evolution of targeted drugs has accelerated the need for equally sophisticated diagnostics. Therapies directed at EGFR, HER2, PD‑L1, BRAF, and ALK have demonstrated that biomarker‑matched treatment can dramatically improve survival. Today, the landscape is expanding to include antibody‑drug conjugates, multi‑specific antibodies, and next‑generation immunotherapies. Each innovation requires precise diagnostic tools capable of identifying the patients most likely to benefit.
Recent advances highlight this momentum. Regulatory agencies worldwide continue to approve new CDx assays, including liquid biopsy‑based tests that detect actionable mutations from a simple blood draw. These innovations are reshaping clinical workflows by enabling faster, less invasive, and more comprehensive biomarker assessment.
The Rise of Liquid Biopsy and Multi‑Omics Integration
Traditional tissue biopsies remain foundational, but liquid biopsies are rapidly gaining ground. They allow clinicians to monitor tumor evolution, detect resistance mutations, and adjust treatment strategies in real time. Meanwhile, multi‑omics approaches—integrating genomics, transcriptomics, proteomics, and spatial profiling—are redefining how biomarkers are discovered and validated.
Artificial intelligence is now entering the field, enhancing pattern recognition, improving assay interpretation, and accelerating biomarker discovery. These technologies are paving the way for diagnostics that are not only predictive but also adaptive, evolving alongside the patient’s disease.
Challenges on the Road to Widespread Adoption
Despite remarkable progress, companion diagnostics face several hurdles. Tumor heterogeneity can complicate biomarker detection, while differences in testing platforms may lead to inconsistent results. Economic and regulatory complexities also influence the pace of CDx development, particularly when diagnostics and therapeutics must be co‑developed and approved together.
Yet the direction is clear: the future of medicine will rely on diagnostics that are tightly integrated with therapeutic design, clinical decision‑making, and patient monitoring.
A Future Built on Precision and Partnership
Companion diagnostics are no longer optional—they are central to modern healthcare. As targeted therapies become more sophisticated, CDx will continue to guide treatment selection, predict response, and monitor disease progression. The next generation of diagnostics will be faster, more accurate, and more deeply embedded in clinical practice, ultimately enabling a healthcare ecosystem where every patient receives a therapy tailored to their unique biology.
Picture source: https://ars.els-cdn.com/content/image/1-s2.0-S2329050123001225-fx1_lrg.jpg